Approaches to Learning
Seven Roots of Education
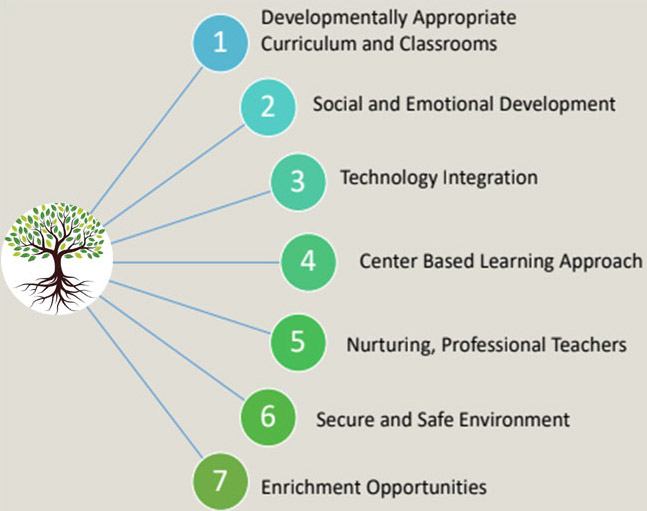
Developmentally Appropriate Curriculum and Classrooms
Social and Emotional Development
Center Based Learning Approach
Technology Integration
Nurturing, Professional Teachers
Secure and Safe Environment
Substance |
Olmesartan |
|---|---|
Brand name: |
Benicar |
Description |
|
Antihypertensive drug |
|
Benicar is used in children from the age of six to normalize high blood pressure due to chronic diseases or taking certain medications |
|
Brands: |
Benicar |
Availability: |
Prescription needed |
Pregnancy: |
Consult a doctor |
Alcohol: |
Doctors do not recommend combining this medicine with alcohol |
Side Effects: |
Cloudy urine |
Interactions: |
Aspirin low strength (aspirin) |
Benicar lawsuit settlement amounts
On August 1, 2017, Daiichi Sankyo reached a 0 million settlement with 2,300 plaintiffs who filed lawsuits over Benicar and related drugs. People who took Benicar filed lawsuits claiming they were unaware it could cause serious gastrointestinal problems. In August 2017, the manufacturer of hypertension drug Benicar announced a 0 million settlement to resolve about 2,300 claims in state and federal courts that the drug caused severe gastrointestinal injuries among patients. Retaliation lawsuits are filed by people who believe they have been the victim of discrimination in the workplace. Benicar is an angiotensin II receptor blocker (ARB), FDA-approved to lower blood pressure associated with hypertension. A portion of this fund will also go to attorney fees and the Claims Administrator.. People filed the first federal Benicar lawsuits in 2014 after the Food and Drug Administration warned that drugs containing olmesartan could cause sprue-like enteropathy, a disease known to cause severe diarrhea and weight loss. Benicar Lawsuit Updates: Filing A Lawsuit. If you have filed a claim that is part of this class action,Read More. Contact us today to find out how we can help you with your lawsuit. To calculate settlement amounts, you must have a reliable total of expenses incurred as a result of the dispute. Kugler entered an order to establish a. In the past, Benicar Lawyers had produced highly informative reports and written legal articles covering all areas of personal injury benicar lawsuit settlement amounts litigation Most lawsuits never make it to trial, and some are settled before the complaint is even formally filed. These lawsuits allege that the manufacturer failed to warn consumers that the drug could cause gastrointestinal problems High blood pressure medications, Benicar and Benicar HCT have been linked to serious gastrointestinal illness known as "sprue-like enteropathy. How much each person receives will be determined by various factors, including: Medical costs related to their Benicar side effects Pain and suffering caused by the drug. Benicar may cause severe gastrointestinal injuries, including sprue-like enteropathy. However, not all of the plaintiffs who file a lawsuit will receive the same amount. Daiichi Sankyo, manufacturer of the blood pressure medication Benicar, released a statement saying it is prepared to settle more than 2,300 lawsuits for 0 million. ” The settlement also says the lawsuits do not have merit. This agreement was the final word on Benicar and a big win for patient justice as a whole. Tracy Reed-Brown had claimed the device she received in 2010 perforated her inferior vena cava and punctured her pancreas, renal vein and aorta Most lawsuits never make it to trial, and some are settled before the complaint is even formally filed. 7 million jury award against Rex Medical over its Option filter. , the manufacturer of Benicar, agreed to pay the United States and state Medicaid programs million to resolve allegations that it violated the False Claims Act by allegedly paying kickbacks to induce physicians to prescribe their drugs, including Benicar. In the past, Benicar Lawyers had produced highly informative reports and written legal articles covering all areas of personal injury litigation.. In the past, Benicar Lawyers had produced highly informative reports and written legal articles covering all areas of personal injury litigation IVC Filter Settlements and Verdicts. The largest IVC filter verdict to date is a . Benicar lawsuits led to a 0 million settlement. Resolved Prior results do not guarantee or predict a similar outcome in any future matter The Benicar lawsuit claimed the makers of Benicar failed to warn doctors and patients that the drug could cause sprue-like enteropathy. Benicar, a olmesartan angiotensin II receptor blocker (ARB), is a medication approved for the treatment of high blood pressure alone or with other antihypertensive agents. ” Benicar Lawsuit Updates: benicar lawsuit settlement amounts Filing A Lawsuit. Benicar has not been taken off the market ANSWER. Food and Drug Administration in 1995. It is estimated that over 11 million patients have been. As a result, new cases are no longer being reviewed by Saiontz & Kirk, P.
What Is Benicar 20mg Used For
According to the FDA safety report: “Olmesartan medoxomil is an angiotensin II receptor blocker (ARB) approved for the treatment of high blood pressure, alone or with other antihypertensive agents, and is one of eight marketed ARB drugs. Sprue-like enteropathy has not been detected with ARB drugs other than olmesartan. The Benicar settlement totals 8 million and covers more than 2,200 cases. The Benicar lawsuit claimed the makers of Benicar failed to warn doctors and patients that the drug could cause sprue-like enteropathy. In addition to a sprue-like enteropathy warning, benicar lawsuit settlement amounts the Benicar label also warns against taking the medication while pregnant as kannst du zocor rezeptfrei kaufen the drug may “cause injury of death to the developing fetus. Daiichi Sankyo reached a 0 million settlement to resolve Benicar lawsuits. Recently, drug makers Daiichi Sankyo and Forest Laboratories have agreed to pay a 0 million Benicar settlement to resolve 2,300 lawsuits regarding these side effects. Cases accusing the Japanese The first lawsuits over Benicar were filed in 2014 and the cases. Call Roden Law today at 1-844-RESULTS for your free legal consultation. The suits involved Benicar and its sister drugs Azor and Tribenzor, all of which benicar lawsuit settlement amounts contain olmesartan as an active ingredient The Benicar lawsuit claimed the makers of Benicar failed to warn doctors and patients that the drug benicar lawsuit settlement amounts could cause sprue-like enteropathy. In 2017, Benicar manufacturer Daiichi Sankyo and co-promoter Forest Laboratories agreed to a 0 million settlement to compensate nearly 2,000 patients for their injuries. A portion of this fund will also go to attorney fees and the Claims Administrator According to the Justice Department, Daiichi Sankyo Inc. Important Benicar Settlement Information In August 2017, a 0 million settlement was reached with Daiichi Sankyo on behalf of individuals who filed lawsuits after suffering sprue-like enteropathy and other gastrointestinal issues from Benicar, Benicar HCT. This can include instances where a person feels they were fired unfairly or that they have experienced a negative influence on the way. And this page is provided for informational purposes only Daiichi Sankyo on Tuesday said it has agreed to pay up to 0 million to settle some 2,300 U. By blocking angiotensin receptors, which can cause blood vessels to constrict, Benicar keeps blood vessels dilated and helps treat high blood pressure The Benicar lawsuit claimed the makers of Benicar failed to warn doctors and patients that the drug could cause sprue-like enteropathy. Benicar was approved by the FDA in 2002 and is distributed by Daiichi Sankyo of Forest Laboratories. A portion of this fund will also go to attorney fees and the Claims Administrator Benicar lawsuits led to a 0 million settlement. In May 2012, the Mayo Clinic published a study of 22. " Benicar's manufacturer, Daiichi-Sankyo has faced numerous Benicar lawsuits for injuries caused by the medication. A further two billion dollars was awarded to a husband and wife who developed non-Hodgkin’s lymphoma that they linked to using roundup, although the amount was reduced to .Enrichment Opportunities
Copyright © 2022 The Sapling School Web.com. All Rights Reserved.
Schedule a Tour
